KaLupso, the turnkey solution for in-house nca 177Lu manufacturing
Contact our expertsMorpho Pharma provides the first fully automated and compact equipment for simple, cost effective and high quality nca 177Lu production.
In-house production of Lu-177 allows to better control the supply chain of this key active ingredient and reduces the risk of backorder
Thanks to its small footprint, the KaLupso equipment can be installed in most standard chemistry hot cells
Accelerate tech transfer and reduce operator time with the first 100% automated and easy-to-use equipment
The KaLupso equipment produces high quality nca Lu-177 that meets industry standards
Adjust the Lu-177 production to your needs depending on batch number/week and target size
The Kalupso equipment consists of a dissolution unit, a chromatography unit and a software that drives the process.
Dissolution Unit
crushes the quartz target and dissolves the irradiated Yb-176 powder. The unit has been created with a focus on chemical and radiation safety
Chromatography Unit
purifies Lu-177 from Yb-176 and other impurities. The radiochemical separation is monitored in real-time via online gamma-detection. The column docking system allows quick and drip-free column removal
User-friendly software
The KaLupso proprietary software enables to set, monitor, and record all process parameters. The production process is automated and requires minimal hands-on time.
nca
nca Lu-177 is produced through the indirect route by neutron irradiation of enriched Ytterbium-176 targets in a nuclear reactor. The Yb-176(n,γ)Yb-177 reaction produces Yb-177 that rapidly decays into carrier-free Lu-177, commonly referred to as non-carrier added 177Lu, due to the absence of Lutetium isotopes in the target resulting in a high specific activity.
Lutetium-177 must then be isolated from the irradiated Yb-176 target by radiochemical separation and purified from any impurities to meet the high purity requirements of the FDA and European Pharmacopeia.
Thanks to its flexible design, KaLupso is suitable for Tb-161 production.
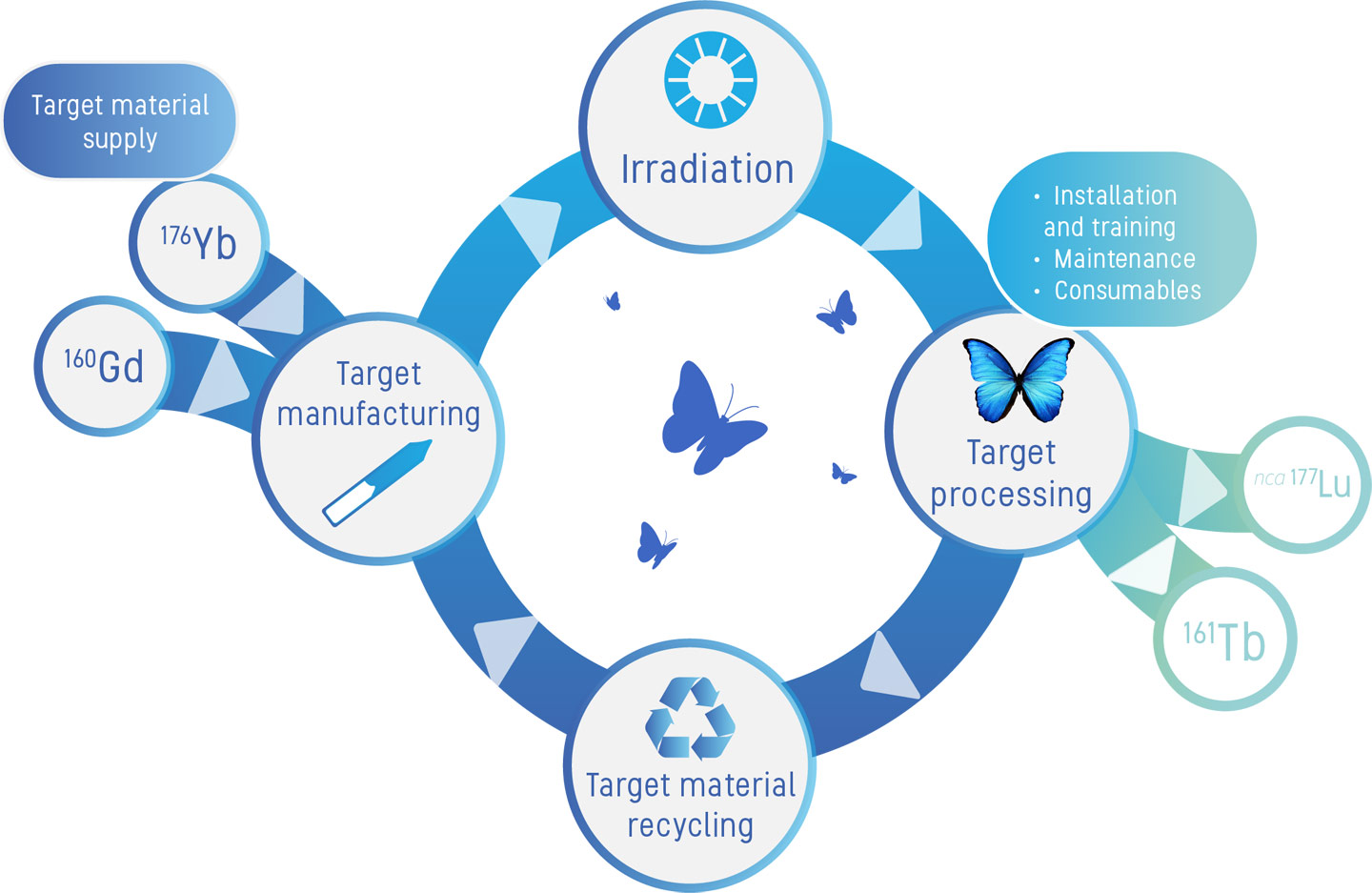
We offer a wide range of services to assist our partners in navigating the complex upstream supply chain of reactor-made medical isotopes and the downstream target recycling process.
- target material supply
- target manufacturing
- irradiation
- target processing: installation & training, maintenance and consumables
- target material recycling